Chemical Synthesis and Reactions of Furan: Furan was first reported by Heinrich Limpricht in 1870. It is a colorless, inflammable, volatile, liquid with a boiling point of 31-32°C. The numbering and structures of furans are as follows:
Chemical Synthesis of Furan
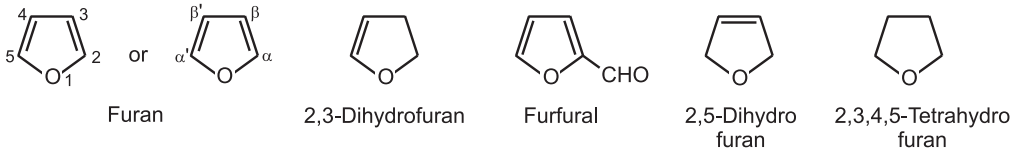
(1) The vapor phase decarboxylation of furfural in the presence of palladium and charcoal gives furan.
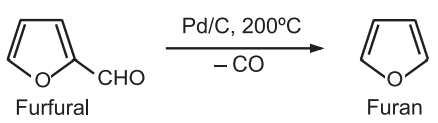
(2) 1,3-Butadiene can be converted to furan by copper-catalyzed oxidation.
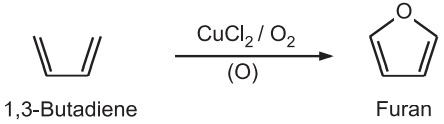
(3) Paal-Knorr Synthesis: Under non-aqueous acidic conditions, 1, 4 – diketones undergo cyclization followed by dehydration to give furans.
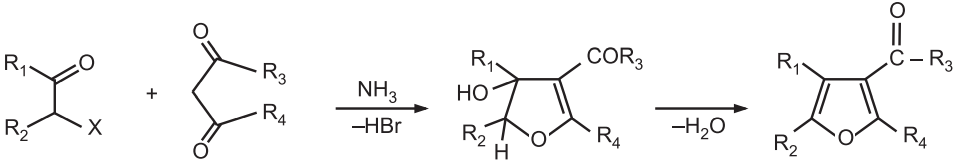
(4) Fiest-Binary Synthesis: It is a condensation reaction between an α – halo ketone with a β – keto ester (or a β – diketone) in the presence of a base like ammonia or pyridine.
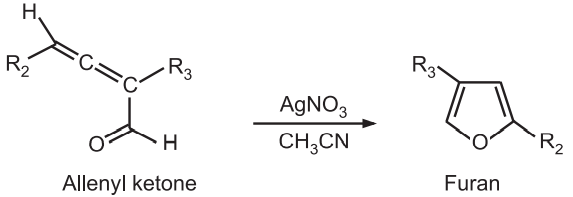
(5) Allenyl ketones undergo cyclization in the presence of silver nitrate or silver boron tetrafluoride in CH3CN to give furans.
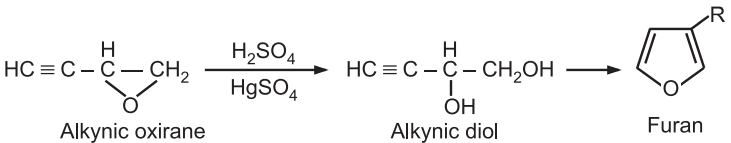
(6) Ring expansion: Alkynic oxiranes when treated with sulfuric acid and mercury sulfate, undergo ring expansion to produce furans.
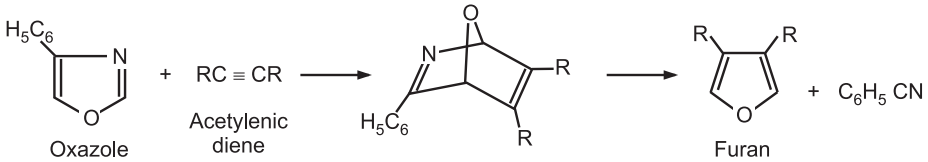
(7) From other heterocycles: Oxazoles undergo Diels-Alder cycloaddition reaction with acetylenic dienophiles. The resulting product provides furan upon loss of nitrile.
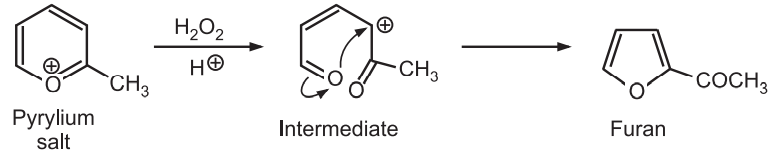
(8) From Ring contraction: Oxidative ring contraction of pyrylium salts with aqueous hydrogen peroxide and perchloric acid leads to the formation of 2-acylfurans.
Chemical Reactions of Furan
Furan is a π-excessive heterocycle and hence prefers electrophilic substitution reactions. At the same time, it behaves chemically as a typical diene and exhibits greater reactivity towards addition reactions.
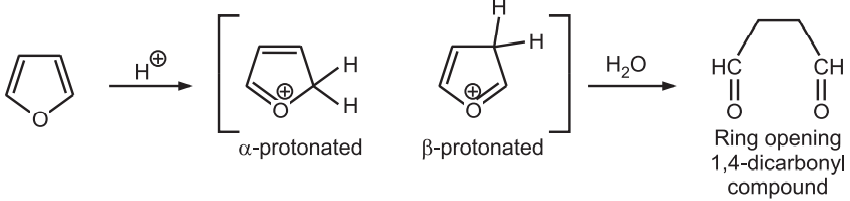
(1) Protonation: Furans substituted with electron-withdrawing substituents are stable towards acid. However, the presence of electron-releasing substituents on furan leads to the generation of reactive electrophiles during protonation which activates polymerization and the ring-opening reactions.
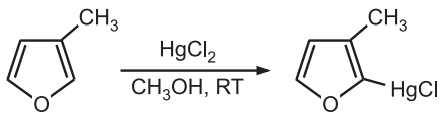
(2) Mercuration: Furan readily undergoes mercuration.
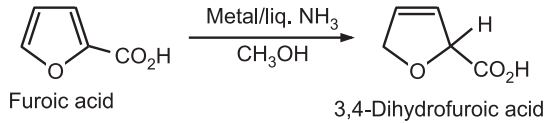
(3) Reduction: Simple furan is difficult to reduce to tetrahydrofuran, without ring opening. Furoic acid can be reduced to a dihydro derivative.
(4) Electrophilic Substitution:
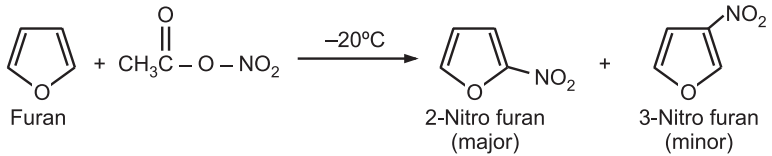
(i) Nitration: Furan is nitrated with a mild nitrating agent, acetyl nitrate, at a low temperature.
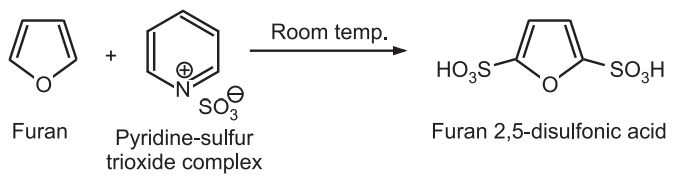
(ii) Sulfonation: Furan is sulfonated with the complex of sulfur trioxide and pyridine or dioxane to give 2, 5 – disubstituted furan even at room temperature.
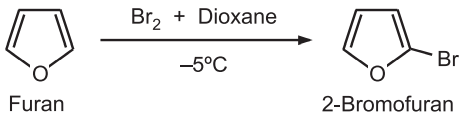
(iii) Halogenation: The high reactivity of furan with chlorine and bromine at room temperature results in polyhalogenated products even at room temperature. Hence, milder conditions are required to yield mono-chloro or mono – Bromo furans. Bromination of furan in DMF or dioxane at –5°C provides 2-bromofuran.
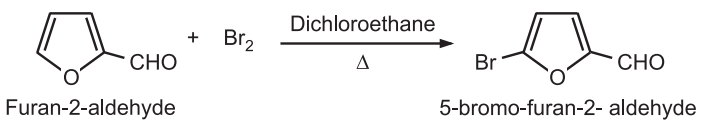
Furan substituted with an electron-withdrawing substituent at position-2 generally provides a 5-Bromo derivative.
(iv) Alkylation: Furan does not undergo Friedel-Crafts alkylation due to its acid sensitivity. Furan can be alkylated at position-2 using alkene in the presence of mild catalysts like phosphoric or boron trifluoride.
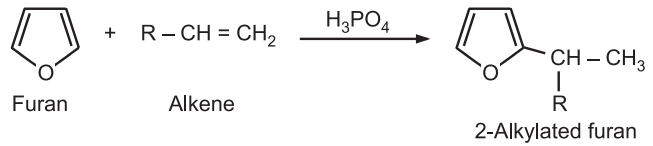
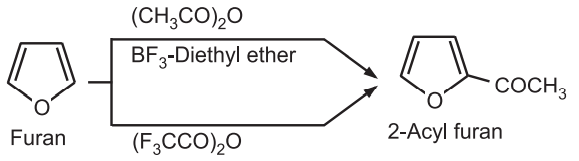
(v) Acylation: The acylation of furan with an acid anhydride or acid halides normally requires mild catalysts like phosphoric acid or boron trifluoride. No catalyst is required if trifluoroacetic anhydride is used as acylating agent.
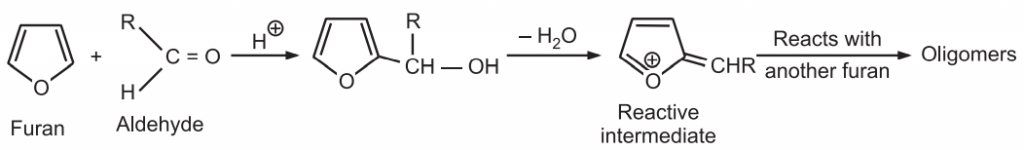
(5) Condensation with aldehydes and ketones: In this acid-catalyzed reaction, furan condenses with aldehydes to produce a mixture of oligomers. A macrocycle is obtained by condensation of furan with acetone.
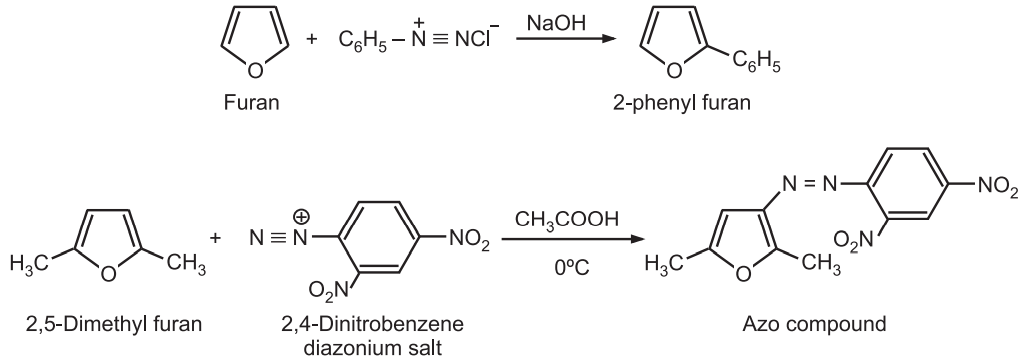
(6) Reaction with diazonium salts: Furan reacts with a benzene diazonium salt to give 2-phenyl furan rather than the formation of an azo compound.
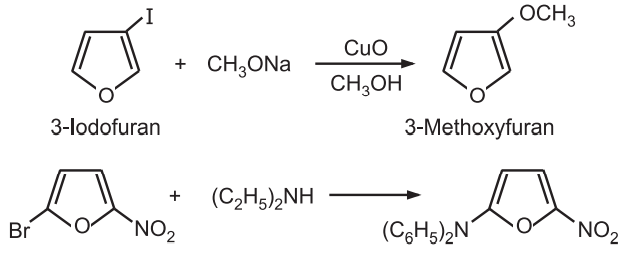
(7) Reactions with nucleophilic reagents: Halofurans show more reactivity towards nucleophiles than simple furan. The presence of electron-withdrawing substituents (nitro, carboxy, or carboalkoxy) in halofurans further increases their reactivity.
(8) Oxidation: Ring-opening occurs when furan is treated with sodium hypochlorite, hydrogen peroxide, or meta chloroperbenzoic acid.
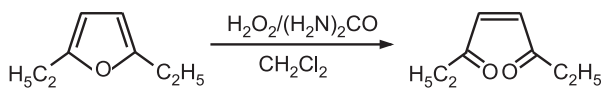
Applications in Drug Synthesis
Furan is an important scaffold present in drugs like ranitidine (antiulcer), nitrofurazone (anti-bacterial), ascorbic acid (vitamin C), and many natural terpenoids.
Make sure you also check our other amazing Article on : Chemical Synthesis and Reactions of Pyrrole
