Drug Packaging: Merely producing a good quality drug product is not enough; it must be packed in a system that ensures the drug retains its activity, efficacy, and safety. The packaging used must not harm the drug product; at the same time, it is also important to ensure that the product does not cause damage to the packaging.

Functions of Packaging:
Table of Contents
The packing material used to deliver a product has several functions:
1. Containment of product is the basic function. The packaging must be effective enough to hold the product without leaking and not cause diffusion of the product.
2. Packaging gives protection against external environmental conditions that may affect the strength, identity, purity, and quality of the product.
3. It helps to identify the product by name and provides all necessary information about the product.
4. Packaging permits the easy use or administration of the drug product.
Classification of Pharmaceutical Packaging:
Packaging materials may be classified into three categories:
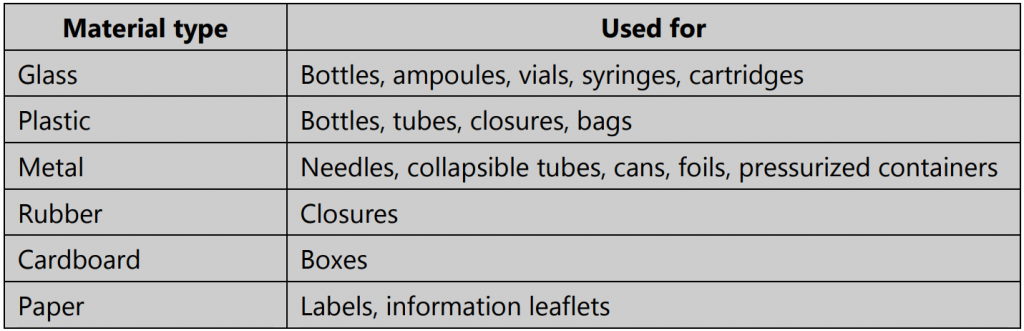
1. Primary packaging which first covers the product and holds it – the parts that come into direct contact with the product. Examples – bottle, ampoule, ointment tube, etc.
2. Secondary packaging surrounds the primary packaging. Examples – box, carton, injection tray, etc.
3. Tertiary packaging that is used for transportation in bulk. Examples – barrel, crate, etc.
Quality Control of Packaging Materials:
The physical and chemical characteristics of packaging material are crucial in determining if it will be able to provide adequate containment and protection to the drug product. Packaging material must be designed keeping in mind the nature of the drug product. QC testing of packaging materials is to be done when the materials are received from the vendor; during the packing process and at the end of packing too, samples are drawn to test the completeness and integrity of the final pack.
Parameters Tested:
Packaging materials are tested for their appearance, dimensions, compatibility, chemical composition, and customer usability. Tests are performed to check the suitability of:
(a) Material of primary packaging – chemical tests, mechanical tests, and environmental tests.
(b) Final pack in secondary packaging – mechanical tests and environmental tests.
Some Important Chemical Tests:
Tests for glass containers:
- Water attack test
- Hydrolytic resistance test
- Powdered glass test
- Light transmission test
- Arsenic test
- Internal bursting pressure test
- Thermal shock test
- Annealing test
- Vertical load test
- Autoclaving
Ampoule sealing tests:
- Appearance
- Quality of seal
- Sealed ampoule length
- Head space oxygen
Plastic containers testing:
- Physicochemical tests on aqueous extracts
- Heavy metals test
- Non-volatile residue
- Biological in-vivo test
- Haemolytic effect of aqueous extracts
- Acute systemic toxicity
Rubber closure testing:
- Self sealability test
- Fragmentation test
Collapsible tubes testing:
- Leakage test
- Lacquer compatibility test
- Lacquer curing test
Strip and blister packs testing:
Paper and board tests:
- Paper and board density
- Moisture content
- Air permeability
- Folding endurance
- Grammage
- Tear strength
- Tensile strength
- Burst strength
- Puncture resistance
- Cobb test for water absorbance
- Creasability
- Stiffness
- Rub resistance
- Ink absorbency
- Brightness
- Roughness/smoothness
Tests on cartons:
- Compression
- Coefficient of friction
- Carton opening force
- Crease stiffness
- Joint shear strength
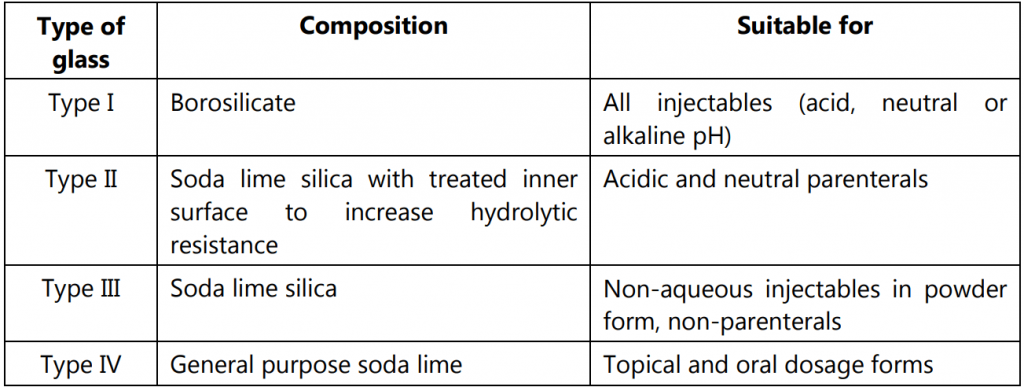
Types of glass containers used in the pharmaceutical industry
Make sure you also check our other amazing Article on : Out of Specification Results