Isolation of Citral: Citral occurs abundantly in lemongrass oil Cymbopogan flexuosus (Nees) Stapf and Cymbopogan citrates (DC) Stapf belongs to the family Graminae. Lemongrass contains about 75 to 85 percent of citral in their oil. It also occurs in verbena oil, lemon, lime, orange, and ginger root. Citral is also present in other species like Ocimum pilosum (contains 35 percent), Liptospermum citratum, Eucalyptus staigeriana, and other citrus species.
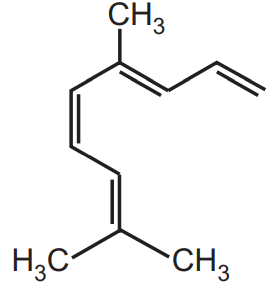
Isolation of Citral
The lemongrass oil is shaken with 5 percent sodium bisulphate solution for 25 to 30 minutes. The resultant is first separated on the Buchner funnel and washed with solvent ether or ethanol. The crude citral is regenerated by decomposing the sodium bisulphate adducts with dilute sodium hydroxide solution. Pure citral is obtained by distillation of crude citral under reduced pressure. Its boiling point is 92 to 93°C.
Separation of Geranial (Citral-a) and Neral (Citral-b):
It was observed by Tiemann that geranial (Citral-a) is obtained free from neral (Citral-b) during the regeneration process from bisulphate adduct. The crystalline sodium bisulphate adduct of Geranial or Citral-a is sparingly soluble in water whereas Neral or Citral-b adduct is readily soluble in water. Tiemann observed that neral is isolated from citral (mixture) by shaking it with an alkaline cyanoacetic acid solution (NC.CH2.COOH) for a short period when geranial reacts at a much faster rate with acid than neral. Citral-a have melting point 108- 110°C and the Citral-b melting point is 96°C.
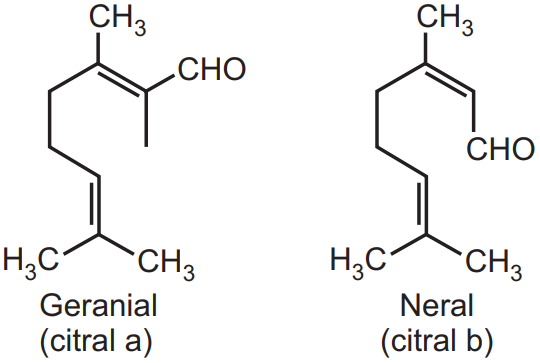
Identification Test of Citral
- Citral is very sensitive to oxidizing agents (even exposure to air) and yields linalool which have an intense yellow colour.
- Geranial with Tollen’s reagent (ammoniacal silver nitrate) gives geranic acid (C19H15COOH).
- Geranial upon hydrogenation with sodium amalgum in acidic solution gives citronellal and citronellol.
- Geranial gets converted into ρ-cymene on treatment with potassium bisulphate or dilutes sulphuric acid.
- When citral interacts with acetone, it forms pseudo-ionone or Ψ–ionone. The aliphatic ketone pseudo-ionone undergoes cyclisation with a variety of reagents like sodium acetate, concentrated sulphuric acid, formic acid, dilute mineral acid etc and yields β-ionone and α-ionone.
Make sure you also check our other amazing Article on : Isolation of Menthol
