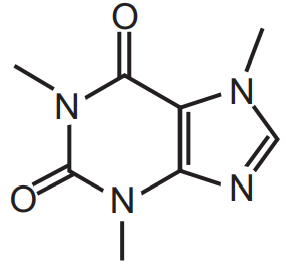
Caffeine can be obtained from different sources like Tea (Camellia sinensis, Family-Theaceae), coffee (Coffea arabica, Family- Rubiacea), cacao (Theobroma cacao Family-Malvaceae), cola (Cola acuminata and Cola nitida Family- Malvaceae), and others. Caffeine can be extracted from tea sweepings, coffee roasters, tea dust, or tea. Caffeine is white in colour or white powder. It is anhydrous or on hydration, only one molecule of water will be attached. It is bitter, needle shape, sublimined crystals. In the presence of salicylate, citric acid, benzoates its solubility in water increases very much.

Production of Caffeine
Caffeine can be extracted from the tea. Around 50 grams of tea powder are taken and extracted with alcohol for around 6 hours in a Soxhlet extractor. The extract is transferred into a porcelain dish that already contains magnesium oxide (30 grams in 200 ml water). Heat it on a steam bath with continuous stirring and evaporate it. Boil the obtained residue three times with 100 ml water. Filter it in the hot condition through the Buchner funnel. Add the 10 percent sulphuric acid (30 ml) to the combined filtrate. Concentrate the above filtrate to one-third of the volume. Filter the solution during hot and extract it 5 times with chloroform (25 ml). Add sodium hydroxide (1 percent, few ml) in chloroform extract so that the decolourisation takes place. An equal quantity of water adds to this and separates the chloroform layer. Evaporate the chloroform extract to obtain crude caffeine which can be recrystallized with hot water. It shows the positive murexide test in which caffeine is treated with a few drops of nitric acid in a porcelain dish and evaporates it to dryness. Add ammonium hydroxide (a few drops) in this residue then purple colour is obtained.
It can be also extracted by supercritical fluid extraction technique by using liquefied carbon dioxide. Liquefied carbon dioxide absorbs the caffeine through moist coffee. Then it is passed through another pressurized vessel containing absorbing media like resin or activated carbon which retain the caffeine. Separate this caffeine by extracting it with chloroform.
Estimation
Dissolve the accurately weigh 0.18 gm powder in 5 ml of anhydrous glacial acetic acid with warming. If caffeine powder is present in hydrated form dehydrate it by drying at 100 to 105°C. Cool the solution and then add 20 ml toluene and 10 ml acetic anhydride. Perform the non-aqueous titration. Determine the endpoint potentiometrically. Perform blank determination to make the necessary correction.
Factor – each ml of 0.1 M perchloric acid ≡ 0.01942 gm of C8H10N4O2 (Caffeine)
Utilization
It is CNS stimulant drug with the category of methylxanthine. It is a very popular psychoactive drug without any legal foundation. Caffeine citrate comes under essential medicines in the WHO list. It can be used for bronchopulmonary dyspepsia. It also develops in a mild form of drug dependence. It can be used as a dietary supplement.
Make sure you also check our other amazing Article on : Sennoside