Pulmonary Drug Delivery System: The respiratory tract is one of the oldest routes used for the administration of drugs. Over the past decades, inhalation therapy has established itself as a valuable tool in the local therapy of pulmonary diseases such as; asthma and COPD (Chronic Obstructive Pulmonary Disease).This type of drug application in the therapy of this disease is a clear form of targeted drug delivery.
The popularly marketed products are inhalation aerosol products for local pulmonary effects.
The drug used for asthma and COPD, e.g. β 2 agonists such as; Salbutamol (albuterol), Terbutaline formoterol, corticosteroids such as Budesonide, Flixotide or Beclomethasone and mast cell stabilizers such as Sodium cromoglycate or nedocromil.
The latest and probably one of the most promising applications of pulmonary drug administration is
- It’s used to achieve systemic absorption of the administered drug substances.
- Particularly for those substances that exhibit a poor bioavailability when administered by the oral route, e.g. peptides or proteins, the respiratory tract might be a convenient port of entry.
Anatomy of Respiratory Tract
Table of Contents
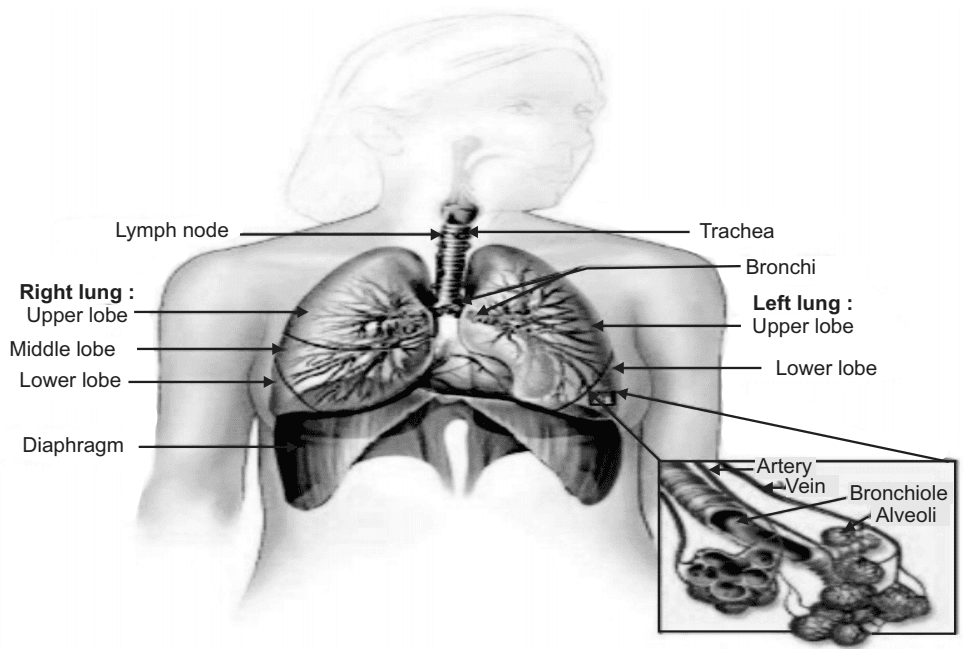
The human respiratory system is a complicated organ system of very close structure-function relationships.
The system consisted of two regions:
- The conducting system
- The respiratory region
- The airway is further divided into many folds: nasal cavity and the associated sinuses, and the nasopharynx, oropharynx, larynx, trachea, bronchi, and bronchioles.
- The respiratory regions consist of respiratory bronchioles, alveolar ducts, and alveolar sacs.
- The human respiratory tract is a branching system of air channels with approximately 23 bifurcations from the mouth to the alveoli. The major task of the lungs is gas exchange, by adding oxygen to and removing carbon-di-oxide from the blood passing through the pulmonary capillary bed.
Advantages of Pulmonary Drug Delivery System
- It is needle-free pulmonary delivery.
- It requires a low fraction of oral dose.
- Pulmonary drug delivery has very negligible side effects since the rest of the body is not exposed to the drug.
- The onset of action is very quick with pulmonary drug delivery.
- Degradation of the drug by the liver is avoided in pulmonary drug delivery.
Disadvantages of Pulmonary Drug Delivery System
- Stability of drug in-vivo.
- Transport.
- Targeting specificity.
- Drug irritation and toxicity.
- Immunogenicity of proteins.
- Drug retention and clearance.
The formulation approaches are as follows:
- Pulmonary delivered drugs are rapidly absorbed except large macromolecules drugs, which may yield low bioavailability due to enzymatic degradation and/or low mucosal permeability.
- The pulmonary bioavailability of drugs could be improved by including various permeation enhancers such as; surfactants, fatty acids and saccharides, chelating agents, and enzyme inhibitors such as protease inhibitors.
- The most important issue is the protein stability in the formulation: the dry powder formulation may need buffers to maintain the pH, and surfactants such as; Tween to reduce any chance of protein aggregation. The stabilizers such as; sucrose are also added in the formulation to prevent denaturation during prolonged storage.
- Pulmonary bioavailability largely depends on the physical properties of the delivered protein and it is not the same for all peptide and protein drugs.
- Insulin liposomes are one of the recent approaches in the controlled release aerosol preparations. Intratracheal delivery of insulin liposomes (dipalmitoylphosphatidylcholine: cholesterol, 7:2) has significantly enhanced the desired hypoglycaemic effect.
- The coating of disodium fluorescein by hydrophobic lauric acid is also an effective way to prolong the pulmonary residence time by increasing the dissolution half-life. In another method, pulmonary absorption properties were modified for protein/peptide drug (rhGCSF) in conjugation with polyethylene glycol (PEGylation) to enhance the absorption of the protein drug by using intratracheal instillation delivery in the rat.
Aerosols
Aerosol preparations are stable dispersions or suspensions of solid material and liquid droplets in a gaseous medium. The drugs, delivered by aerosols are deposited in the airways by:
- Gravitational sedimentation
- Inertial impaction
- Diffusion
Mostly larger drug particles are deposited by the first two mechanisms in the airways, while the smaller particles get their way into the peripheral region of the lungs by the following diffusion.
There are three commonly used clinical aerosols:
- Jet or Ultrasonic Nebulizers
- Metered Dose Inhalers (MDI)
- Dry Powder Inhalers (DPI)
The basic function of these three completely different devices is to generate a drug-containing aerosol cloud that contains the highest possible fraction of particles in the desired size ranges.
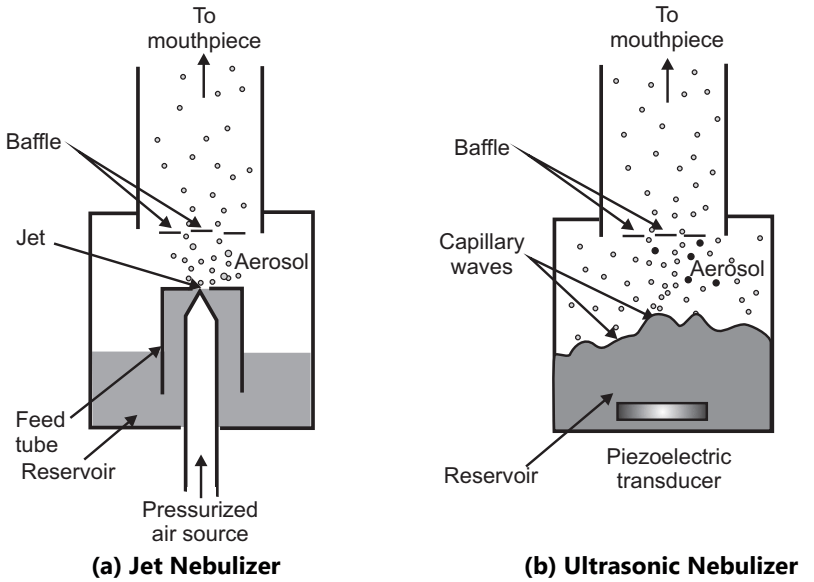
Nebulizers: These are widely used as aerosolized drug solutions or suspensions for drug delivery to the respiratory tract and are predominantly useful for the treatment of hospitalized patients, delivered by the drug in the form of mist. There are two basic types:
- Air jet
- Ultrasonic nebulizers
Metered Dose Inhalers (MDI)
These are used in the treatment of respiratory diseases such as; asthma and COPD. They can be given in the form of suspension or solution. Particle size of fewer than 5 microns is observed here. This is used to minimize the number of administration errors. The main advantage is that it can deliver a measured amount of medicament accurately.
Advantages:
- It delivers a specified amount of drugs.
- Small size and convenience.
- Usually inexpensive as compared to dry powder inhalers and nebulizers.
- Quick to use.
- Multidose capability more than 100 doses available.
Disadvantages:
- Difficult to deliver high doses.
- The remaining amount of doses is not known in MDI.
- Accurate coordination between activation of a dose and inhalation is essential.
Dry Powder Inhalers (DPI)
- DPIs are bolus drug delivery devices that contain the solid drug in a dry powder mix (DPI) that is fluidized when the patient inhales.
- DPIs are typically formulated as one-phase, solid particles blends. The drug with particle sizes of less than 5 µm is used.
- DPIs are a widely accepted inhaled delivery dosage form, particularly in Europe, where they are currently used by approximately 40% of asthma patients.
Advantages:
- Propellant free.
- Less need for patient coordination.
- Fewer formulation problems.
- Dry powders are at a lower energy state, which reduces the rate of chemical degradation.
Disadvantages:
- Dependency on patient’s inspiratory flow rate and profile.
- Device resistance and other design issues.
- Greater potential problems in dose uniformity.
- More expensive than pressurized metered-dose inhalers.
Unit Dose Devices: Single-dose powder inhalers are devices in which a powder-containing capsule is placed in a holder. The capsule is opened within the device and the powder is inhaled.
Multi-Dose Devises: This device is truly a metered-dose powder delivery system. The drug is contained within a storage reservoir and can be dispensed into the dosing chamber by a simple back and forth twisting action on the base of the unit.
Recent Advances in Pulmonary Drug Delivery:
- The Aerogen Pulmonary Delivery Technology.
- The AERx Pulmonary Drug Delivery System.
- The Spiros Inhaler Technology.
- The Direct HalerTM Pulmonary Device Platform.
- Newer Development-Dose Counter Inhalers (DCI).
Make sure you also check our other amazing Article on : Gastro Retentive Drug Delivery System
