The situation shown in Fig.1 describes that free energy is present in the form of tension at the surface. Tension at the surface helps maintain the minimum surface possible. This energy is called surface free energy. Surface free energy can be defined, as the work required in increasing the area by one cm2. The surface free energy can be derived from the following illustration.
ABCD is a three-sided frame with a movable bar CD of length L. A soap film is formed over the area ABCD. Applying a force F to the movable bar, the film stretches downward.
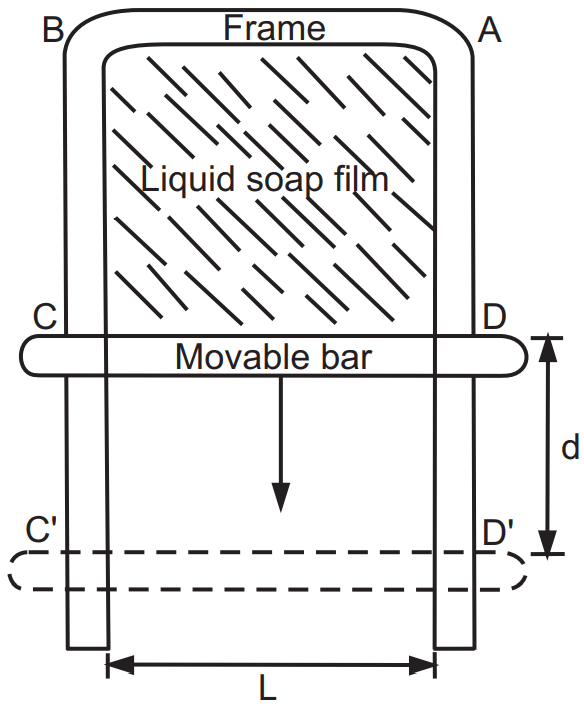
To break the film some force is required. If the applied force is less than what is required to break the film then the film retracts due to surface tension. If the force F is applied on a movable bar CD, it shifts by a distance d to CD. The work done W is expressed as;
W = F × d … equation (1)
While stretching the film the force acts against the surface tension of the liquid as it tries to contract the liquid. The soap film has a liquid-gas interface. The total length of contact of the film is equal to the double length of the bar because the film has two surfaces on either side. Therefore, the force acting on the surface is expressed mathematically as
F = γ × 2L … equation (2)
Substituting values of downward force F, in equation (1), gives;
W = γ × 2L × d … equation (3)
The quantity 2L is equal to the increased surface area ∆A produced by extending the film. Then the equation (3) changes to:
W = γ × ∆A or ∆G = γ × ∆A … equation (4)
where W is work done or increased surface energy expressed in ergs.
In the thermodynamic sense, any form of energy can be split into two factors namely, intensity factor and capacity factor. In film stretching surface tension is the capacity factor. The equation (4) is applied in gas adsorption studies on the solid surfaces, in studying the physical instability of suspensions and thermodynamic instability of emulsions. The dimensional analysis of the work-energy theorem shows that the unit of surface tension (N/m or dyne/cm) is equivalent to J/m2. This means surface tension also can be considered as surface free energy.
Surface Free Energy Measurement:
Following are some methods used to measure the free energy of solid materials.
Dyne Pen Method:
This method involves the use of a set of commercially available felt-tip pens containing a range of inks of known surface tension. One of the pens is used to apply a thin film of ink over an area of the test surface that may be solid or liquid. If the ink film breaks up into droplets in less than two seconds, the process is repeated using a pen with ink having a lower surface tension. This procedure is used to establish the lowest surface tension ink that yields a film that remains intact for at least two seconds. The value of the surface tension of the ink is taken as the surface free energy of the substrate.
Contact Angle Method:
In this method, a drop of liquid of known surface tension is placed on the test surface and then observed through a movable eyepiece. The eyepiece is connected to an electronic protractor, which displays the viewed angle. The construction of the angle is such that while viewing, angle equals the contact angle; the illumination viewed through the eyepiece is maximized. The contact angle and the surface tension of the liquid can then be used to calculate the surface energy of the test surface.
Interfacial Tensiometer Method:
The method involves the use of a tensiometer in which the solid is dipped into and retracted from a liquid of known surface tension. The variation of contact angle with immersion depth is measured and these values are used to calculate the surface free energy of the solid. This method is used for the solids where all exposed faces have the same composition.
Example.1: If the length of the bar is 5 cm and the force required to break a soap film is 0.4 g. What is the surface tension of soap solution? What is the work required to pull the wire by 1 cm?
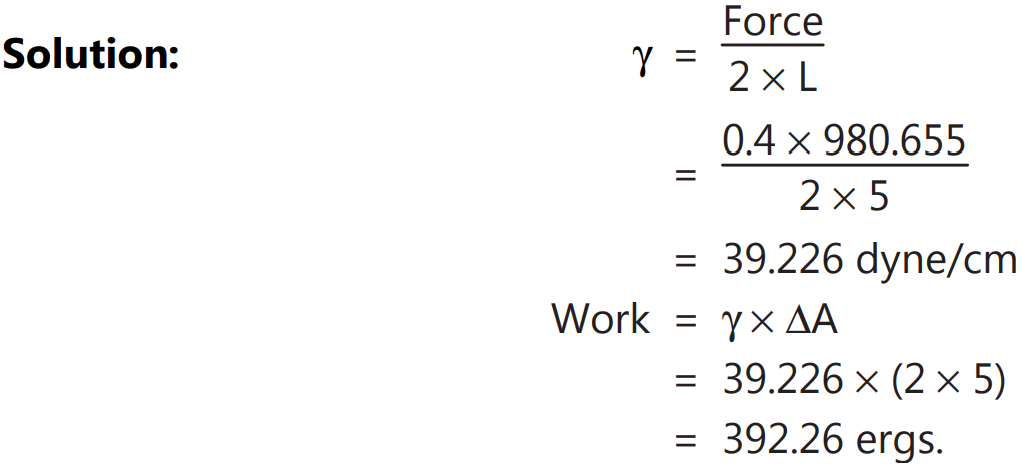
Make sure you also check our other amazing Article on : Surface Tension