Tragacanth Gum: The economical part of the plant is a gum which is a dried gummy exudation and obtained from Astragalus gummier or other species of Astragalus.
Family: Leguminosae.
Distribution: The thorny shrubs of tragacanth normally grow at an altitude of 1000-3000 meters and the primary source is the desert highlands of the northern and western part of Iran. Apart from Iran, it is naturally found in various countries, viz., Iraq, Armenia, Syria, Greece, and Turkey. Very few species of Astragalus are located in India, viz., Kumaon, Garhwal, and Punjab.
Collection of Gum: The collection of gum is described in Fig.1.
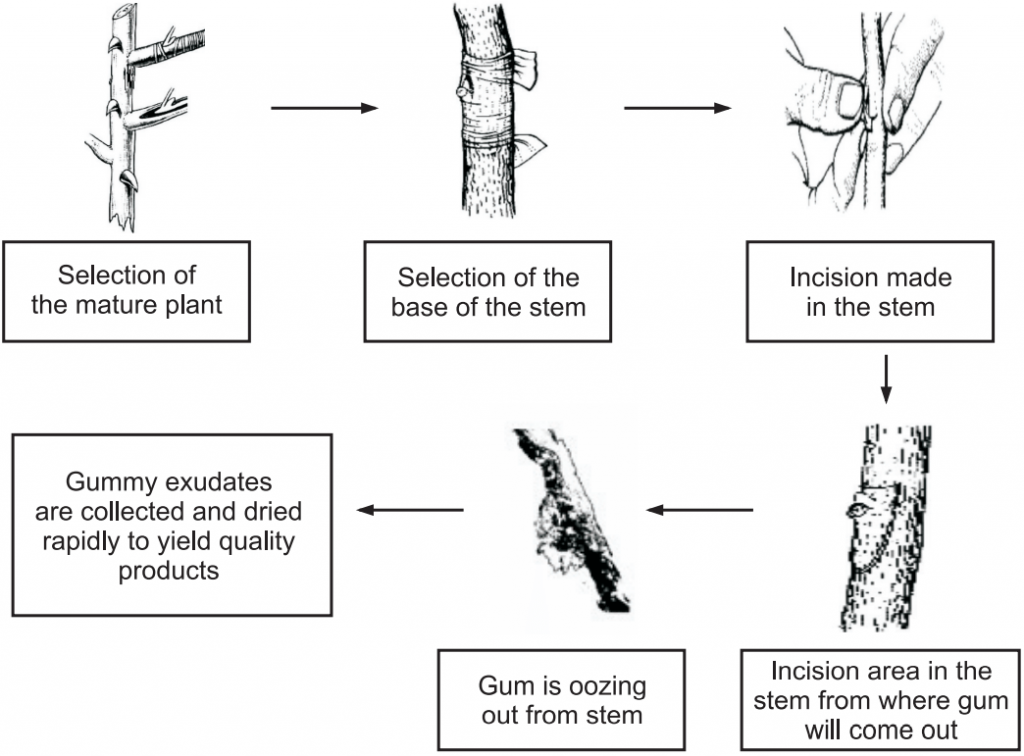
It takes about a week to collect the gum exudates and this process continues periodically.
Properties of Gum:
Table of Contents
- The gum is translucent and the powder is a white or pale yellow, odourless and tasteless.
- Gum looks like a curved or twisted ribbon, like flakes marked with concentric ridges.
- The fracture of the gum is normally short and horny.
- Gum tragacanth is a viscous and water-soluble mixture of polysaccharides.
- It absorbs water to become a gel, which can be stirred into a paste.
- Tragacanthin partially dissolves and partially swells in water yielding a viscous colloid.
Chemical Nature of Gum:
- Tragacanth gum contains linear chains of galacturonic acid in xylose and with varying levels of fructose.
- Tragacanth contains from 20% to 30% of a water-soluble fraction called tragacanthin (composed of tragacanth acid and arabinogalactan).
- It also contains from 60% to 70% of a water-insoluble fraction called bassorin.
- Tragacanth acid is composed of D-galacturonic acid, D-xylose, L-fructose, D-galactose, and other sugars.
- Tragacanthin is composed of uronic acid and arabinose and dissolves in water to form a viscous colloidal solution (sol), while bassorin swells to form a thick gel.
- The gum also contains several carbohydrates like L-arabinose, D-galactose, D-Xylose, L-fructose, L-rhamnose etc.
Chemical Tests for Tragacanth Gum:
- An aqueous solution of Tragacanth + Conc. Hydrochloric acid → Boil → No Red colour forms.
- Sample of Tragacanth + Ruthenium red solution →No Pink colour forms
- An aqueous solution of Tragacanth + Drops of Ferric chloride → Deep yellow precipitate forms.
- Sample solution + Lead acetate → Heavy white precipitate forms
Substituents/Adulterants:
Gum kondagogu is gummy exudate obtained from the tree bark Cochlospermum Gossypium. Dextrin, wheat, and corn starch, etc. are used as substituents for tragacanth gum. Generally, Karaya gum or sterculia gum, or Indian tragacanth is used as a substitute for gum tragacanth.
Karaya gum can be identified morphologically, microscopically, and by chemical tests. (a) Indian tragacanth is irregular, striated, pale brownish pieces and has an acid odor. (b) In Karaya gum powder, sclerenchymatous cells are present, whereas these cells are absent in Tragacanth gum powder. (c) Karaya gum is identified with the ruthenium solution test with the formation of red color, whereas this test is negative for tragacanth.
Citral gum obtained from Acacia strobilaceous is used as an adulterant. It can be identified by chemical tests. Powdered Tragacanth is mixed with water, guaiacol, and few drops of hydrogen peroxide. Pure Tragacanth remains colorless, whereas acacia gum forms a brown color mucilage.
Uses of Tragacanth Gum:
Tragacanth is used as an emulsifier, binding agent, and demulcent. Orally, tragacanth is used both for diarrhea and as a laxative. Topically, tragacanth is an ingredient in toothpaste, hand lotions, and vaginal creams, and medicinal jellies like spermicidal jelly. It is used as a binding agent for the preparations of tablets and pills. In foods, tragacanth is used as a stabilizer, thickener, and suspending ingredients in salad dressings, foods, and beverages. The mucilage is used as adhesives.
Mechanism of Action:
Tragacanth contains ingredients that stimulate the movement of the intestines. It acts as a stabilizer by the formation of non-covalent protein-polysaccharide complexes via interactions by the methoxylated galacturonic acid in the soluble part of the gum and by the viscosity increase induced by the insoluble bassorin part.
Make sure you also check our other amazing Article on : Acacia
