Carbohydrates are one of the most important nutrients that support life on earth. From the food we eat to the energy our body uses, carbohydrates play a major role everywhere. They are not only the main source of energy for humans but are also essential for plants and microorganisms. In this article, we will understand the structure, classification, and functions of carbohydrates in simple terms.
What Are Carbohydrates?
Carbohydrates are natural compounds made up of three main elements — carbon (C), hydrogen (H), and oxygen (O). Their basic formula is often written as (CH₂O)n, which means they are composed of carbon and water in a fixed ratio.
They are also known as sugars or saccharides, depending on their complexity. From the sweet taste of fruits to the fiber in vegetables and grains, carbohydrates exist in different forms and serve various functions in living organisms.
Some carbohydrates provide quick energy (like glucose), while others build structural materials (like cellulose in plants).
Classification of Carbohydrates
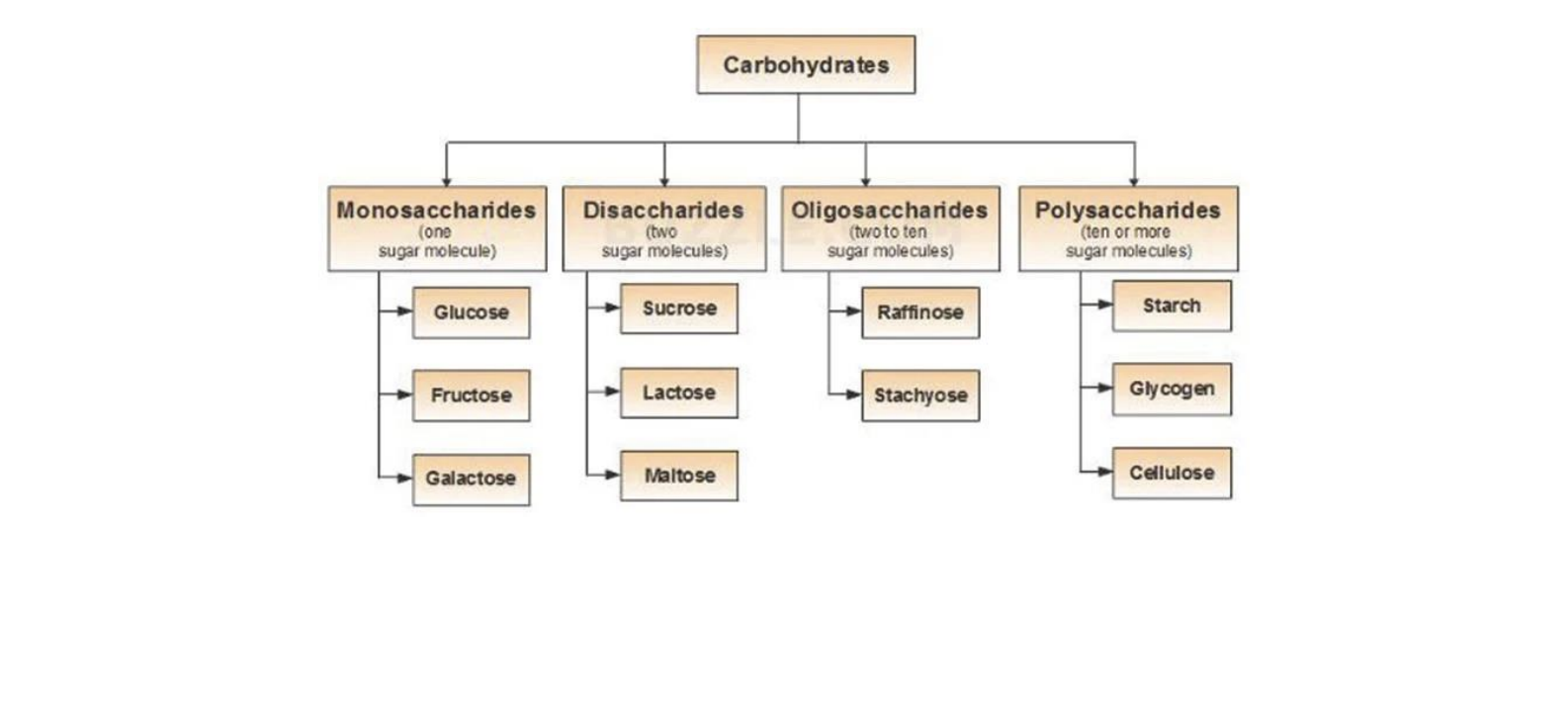
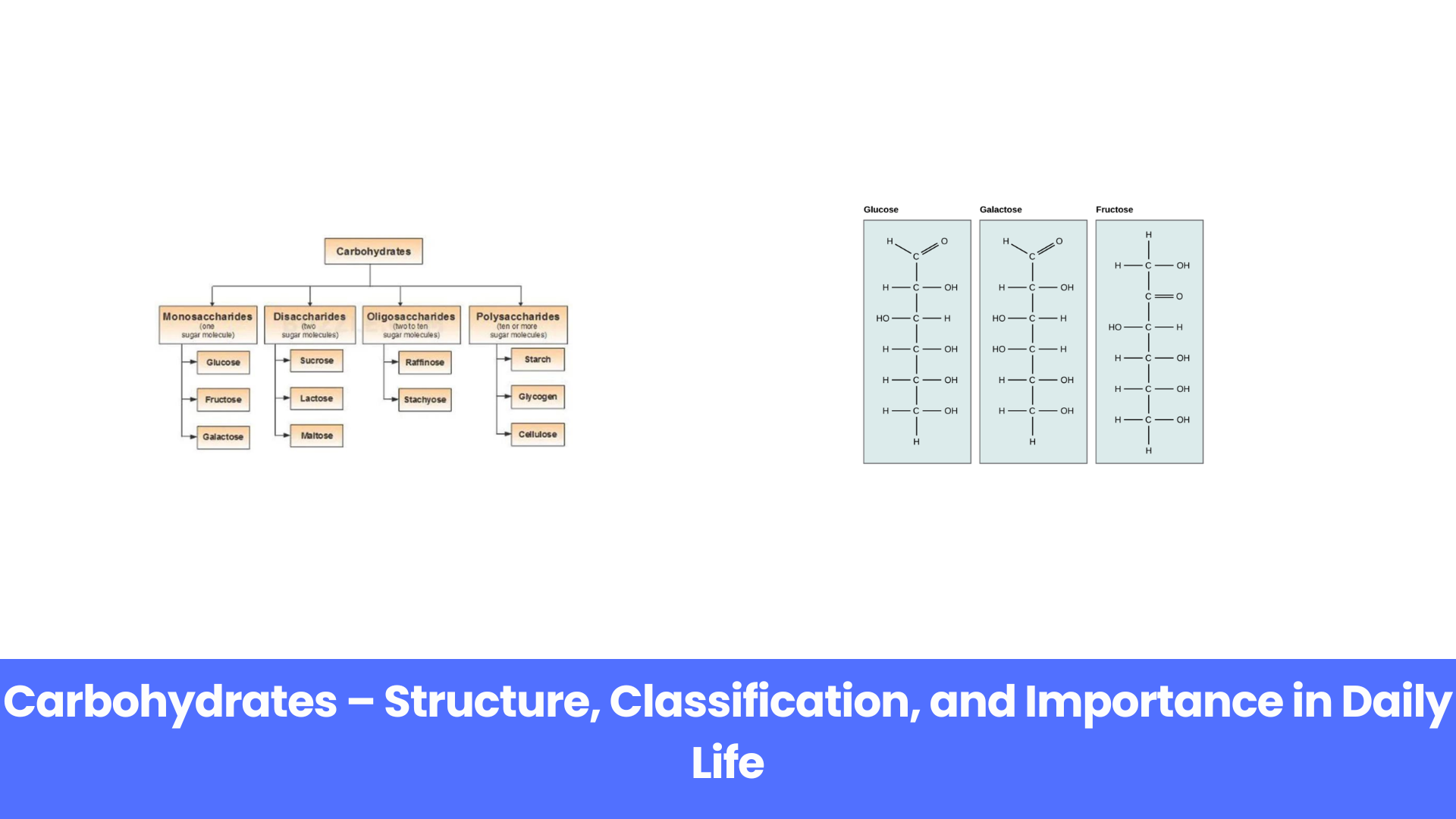
Carbohydrates are classified based on the number of sugar units (monomers) they contain. There are mainly four types of carbohydrates:
- Monosaccharides
- Disaccharides
- Oligosaccharides
- Polysaccharides
Let’s understand each type in detail.
Monosaccharides – The Simple Sugars
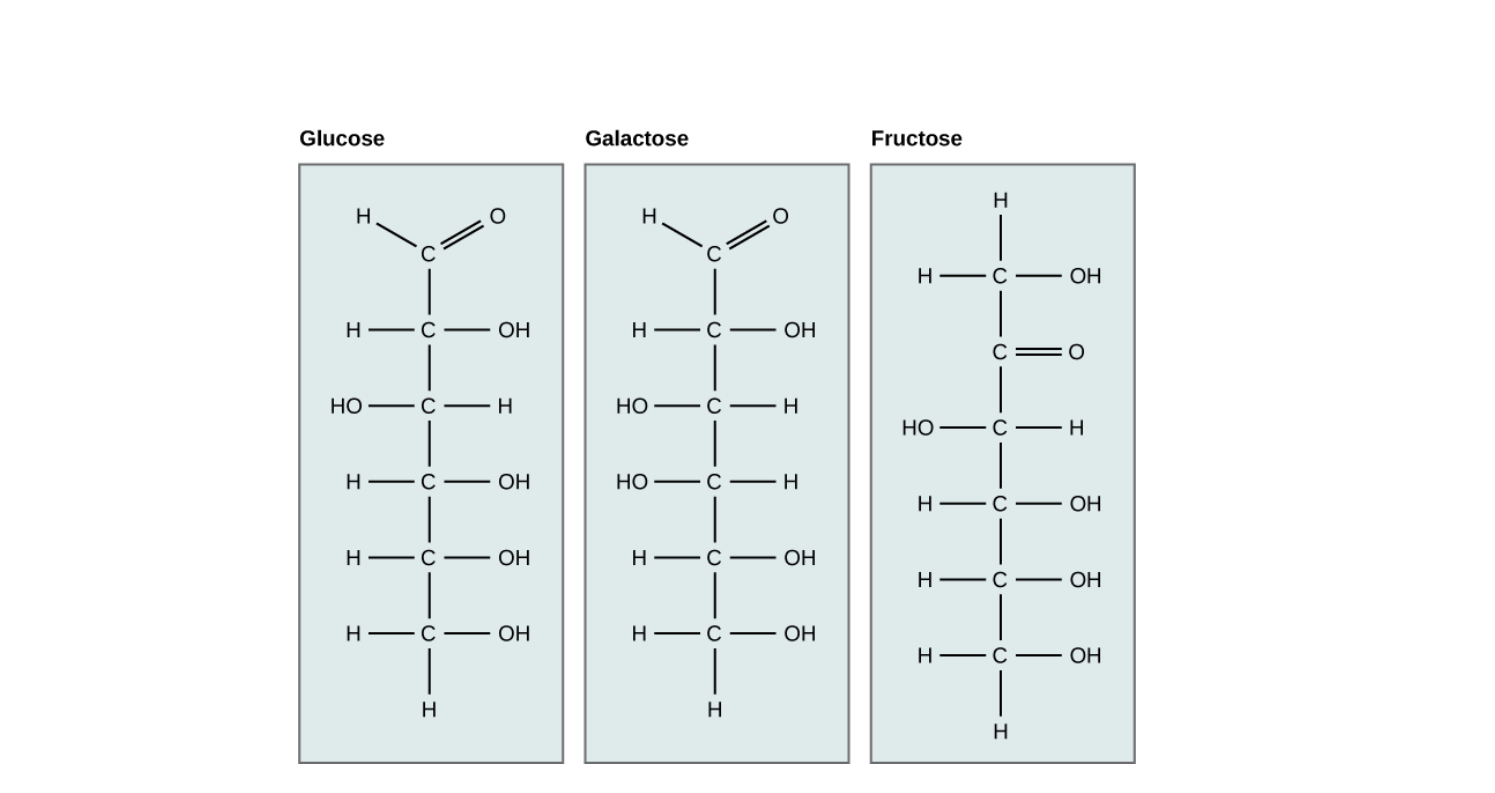
Monosaccharides are the simplest form of carbohydrates. They contain only one sugar unit and cannot be broken down into smaller carbohydrates. They are also called simple sugars.
Examples include glucose, fructose, galactose, and ribose. All these have the same chemical formula (C₆H₁₂O₆) but differ in structure and properties.
Monosaccharides can have three to seven carbon atoms. Based on this, they are called triose (3C), tetrose (4C), pentose (5C), hexose (6C), etc.
- Glucose – The most common sugar that provides energy to our body.
- Fructose – Found in fruits and honey, responsible for their sweetness.
- Galactose – Present in milk and dairy products.
- Ribose – A part of RNA and important for genetic material.
Monosaccharides can exist in open-chain or ring form, and the arrangement of atoms determines whether it is in alpha (α) or beta (β) configuration.
Functions of Monosaccharides:
- Provide instant energy to cells.
- Help in the formation of DNA and RNA.
- Act as building blocks for larger carbohydrates.
- Support normal metabolic functions in the body.
Disaccharides – Double Sugars
Disaccharides are formed when two monosaccharide molecules join together through a glycosidic bond. This bond is formed during a process called dehydration reaction, where one water molecule is removed.
Common examples of disaccharides include:
- Sucrose (glucose + fructose) – Table sugar used in homes.
- Lactose (glucose + galactose) – Found in milk.
- Maltose (glucose + glucose) – Produced during starch digestion.
Disaccharides can be of two types:
- Reducing sugars – Have a free aldehyde or ketone group (e.g., maltose, lactose).
- Non-reducing sugars – No free reactive group (e.g., sucrose).
Functions of Disaccharides:
- Provide a quick energy source.
- Sucrose acts as a transport sugar in plants.
- Lactose gives nourishment to infants.
- Maltose is an intermediate in carbohydrate metabolism.
Oligosaccharides – Short Sugar Chains
Oligosaccharides are made of 3 to 10 monosaccharide units. They are often attached to proteins and lipids in the cell membrane, forming glycoproteins and glycolipids. These combinations help in cell recognition and communication.
There are two main types:
- N-linked oligosaccharides – Attached to nitrogen atom of amino acid asparagine.
- O-linked oligosaccharides – Attached to oxygen atom of amino acid serine or threonine.
Functions of Oligosaccharides:
- Help in cell recognition and immune response.
- Form part of glycoproteins and glycolipids in the cell membrane.
- Play a key role in cell signaling and protection.
- Act as prebiotics, supporting healthy gut bacteria.
Polysaccharides – Complex Carbohydrates
Polysaccharides are long chains of monosaccharides linked by glycosidic bonds. They can contain hundreds or even thousands of sugar units. These are also called complex carbohydrates and serve as energy storage or structural materials.
They are divided into two main groups:
- Homopolysaccharides – Made of one type of sugar (e.g., starch, glycogen, cellulose).
- Heteropolysaccharides – Made of different types of sugars (e.g., peptidoglycan, agarose, glycosaminoglycans).
Examples and Functions:
- Starch – Energy storage in plants. Found in rice, potatoes, and wheat.
- Glycogen – Energy storage in animals, mainly in the liver and muscles.
- Cellulose – Provides strength to plant cell walls; source of dietary fiber.
- Chitin – Found in the shells of insects and crustaceans.
- Agarose – Used in laboratories for making gels.
- Peptidoglycan – Gives strength to bacterial cell walls.
Functions of Polysaccharides:
- Act as energy reserves (starch in plants, glycogen in animals).
- Provide structural support (cellulose in plants, chitin in insects).
- Help in biological processes like cell protection and communication.
- Contribute to joint lubrication (hyaluronic acid in humans).
Conclusion
Carbohydrates are essential for every living organism. From providing energy to forming structural components, their importance cannot be overstated. Whether it is glucose fueling our cells or cellulose forming the structure of plants, carbohydrates are everywhere.
They exist in different forms — monosaccharides, disaccharides, oligosaccharides, and polysaccharides, each performing a unique function. Apart from energy, they play vital roles in immunity, digestion, and cellular communication.
Understanding carbohydrates helps us appreciate how life functions at the molecular level and how our daily diet directly supports our body’s energy and growth.
FAQs
What are the main elements present in carbohydrates?
Carbohydrates are made up of carbon (C), hydrogen (H), and oxygen (O) atoms.
Which carbohydrate is known as blood sugar?
Glucose is known as blood sugar because it circulates in the bloodstream and provides energy to cells.
What is the difference between starch and cellulose?
Both are made of glucose units, but starch is used for energy storage in plants, while cellulose forms the structural part of plant cell walls.
Why are carbohydrates important for the human body?
They are the body’s main source of energy and help in proper brain function, muscle activity, and metabolism.
What are reducing sugars?
Reducing sugars are carbohydrates that can donate electrons in a chemical reaction because they have a free aldehyde or ketone group. Examples include glucose and maltose.
What happens if we eat too many carbohydrates?
Excess carbohydrate intake can lead to weight gain, high blood sugar levels, and an increased risk of diabetes if not balanced with physical activity.