Isolation of Rutin: Rutin is a flavonoid compound that is obtained from citrus fruits. It is also extracted from the leaves of buckwheat Fagopyrum esculentum belongs to the family Polygonaceae. It is used as a capillary fragility factor.
Coarsely powdered drug of buckwheat is first defatted with n-hexane. Then extract the marc with alcohol (78%) for 60 minutes. Filter the solution and evaporate the solvent. The obtained dried residue is dissolved in a sufficient quantity of acetone (30%). Filter the solution and evaporate the filtrate to one-fourth of its original weight. Then add a sufficient quantity of borax solution (5%) until pH 7.5 with continuous shaking. Then add enough quantity of solid sodium chloride with stirring. Filter the solution and acidify it with phosphoric acid to lower the pH 5.5. Stir the solution for 15 minutes and filter it. Wash the residue with sodium chloride (20%) solution. Again filter the solution and evaporate the filtrate to 500°C to one-fourth of its original volume. Then add hydrochloric acid in hot conditions to lower the pH to 1.5. Cool the solution and kept in the refrigerator overnight. Rutin crystals will separate, be collected, and dried.
Melting point: 242°C.
Thin-layer chromatography of Rutin
1% methanolic solution of rutin is spotted on silica gel-G plates. The plates were eluted in a solvent system of n-butanol- glacial acetic acid and- water (3.6:0.5:0.5). The plates were dried and isolated compound detected under UV cabinet at 366nm.
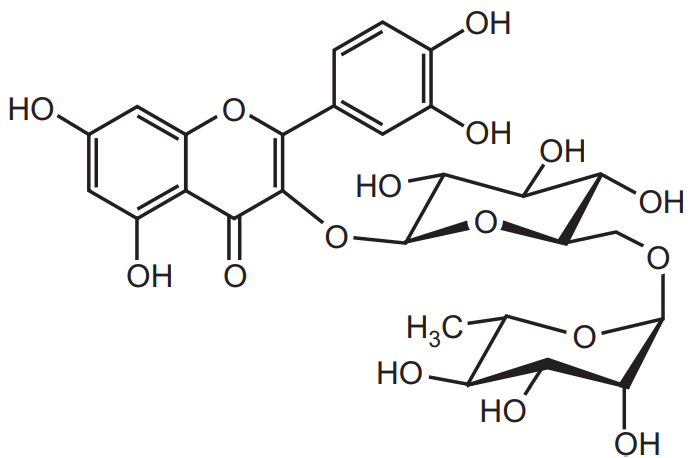
Identification Test of Rutin
- Shinoda test: Add a few drops of concentrated hydrochloric acid or sulphuric acid and magnesium powder in 2 ml of sample solution. An orange, pink, red or purple colour develops.
- Add sulphuric acid in sample solution shows deep yellow colour or orange colour.
- A yellow precipitate is formed when the test solution is mixed with lead acetate.
Make sure you also check our other amazing Article on : Isolation of Caffeine
