Principle for Limit Test for Sulphate
Table of Contents
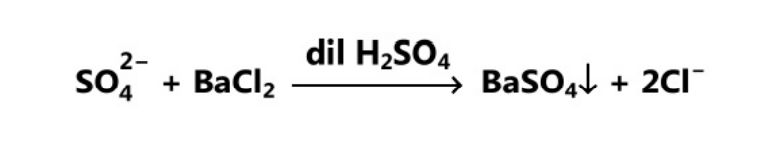
Limit Test for Sulphate is based upon the chemical reaction between barium chloride and soluble sulphate in the presence of dilute hydrochloric acid. The turbidity produced is compared with the standard solution. Barium chloride reagent contains barium chloride, sulphate-free alcohol and a small quantity of potassium sulphate. The inclusion of a small quantity of potassium sulphate in the reagent increases the sensitivity of the test. Alcohol prevents supersaturation and more uniform turbidity develops. If the turbidity produced in the test is more intense than the standard turbidity, then the drug fails the test, otherwise, it passes the test.
Apparatus Required
- Nessler cylinders
- Glass rod
- Stand
Chemicals Required for Limit Test for Sulphate
- Dilute hydrochloric acid
- 0.5 M Barium chloride: 122.1 g of BaCl₂. 2H₂O dissolved in distilled water.
- Barium sulphate reagent containing 0.5 M barium chloride in 1000 ml of water. (This is prepared as follows: Mix 15 ml of 0.5 M BaCl2, 55 ml of water and 20 ml of sulphate free alcohol. Add 5 ml of 0.0181% w/v potassium sulphate. Dilute to 10 ml with water and mix.)
Reaction
Procedure for Limit Test for Sulphate
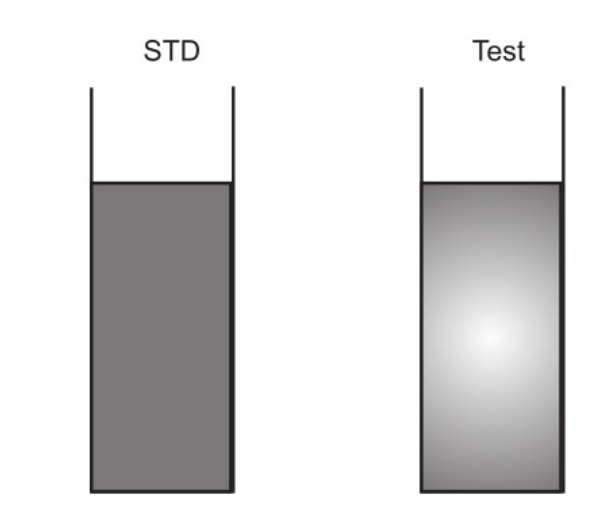
Take two 50 ml Nessler cylinders. Label one as “Test” and the other as “Standard”
| Standard | Test |
| 1. Place 1 ml of 0.1089% w/v solution of K₂SO4 in a Nessler cylinder. | 1. Dissolve the specified quantity of the substance in distilled water and transfer it to the Nessler cylinder. |
| 2. Add 2 ml of dilute. HCI. | 2. Add 2 ml of dilute. HCI. |
| 3. Dilute to 45 ml with water and add 5 ml of barium sulphate reagent. | 3. Dilute to 45 ml with water and add 5 ml of barium sulphate reagent. |
| 4. Stir immediately with a glass rod and allow to stand for 5 minutes. | 4. Stir immediately with a glass rod and allow to stand for 5 minutes. |
| 5. Observe the turbidity developed and compare it with that of the sample. | 5. Observe the turbidity developed and compare it with that of the standard. |
Make sure you also check our other amazing Article on Limit Test for Chloride Limit Test for Arsenic Limit Test for Heavy Metals Limit Test for Lead
