The pharmacy and therapeutic committee (PTC) is a group of medical staff and they perform advisory functions for the safety of patient’s health. The PTC has the power to decide the entry of the new drug into the hospital formulary. Pharmacy and Therapeutic Committee (PTC) generally supervises all facets of drug therapy in a hospital or an institution. The committee usually comprises a committee of healthcare providers composed of physicians, pharmacists, and other health care professionals and they provide the advisory functions and assist in the preparation of policies regarding selection of drug, procurement, dispensing, administering of medications, safety procedures as well as they are involved in various administrative services such as; evaluation of medication use. The committee compares the cost and benefits of each drug and makes a fruitful decision on which drug is suitable and the most efficacious for the respective patient. Physicians and pharmacists both work collaboratively in these committees to implement cost-effective drug regimens for the patients for clinical outcomes. Collaboratively both physician and pharmacist take several initiatives toward patient health. For best clinical output, the committee takes several educational initiatives, training programs, financial incentives, administrative policy to restrict unauthorized practices, use of formularies, and prescribing guidelines. The committees employ an evidence-based approach within health systems/plans by altering/removing old policies with a new one to support in making of decisions for medical staff and patients.
Composition, Organization, and Operation of PTC
Table of Contents
The composition of the pharmacy and therapeutic committee may vary but the mostly following format is suggested for general approval. The PTC may be composed of:
- At least three physicians from various fields like; Internal medicine, Infectious disease, Pediatrics, Surgery, Gynecology. One of the physicians may be appointed as the “Chairman” of the Pharmacy and Therapeutics Committee.
- A senior representative from hospital administration (hospital director) or similar designated person and ex-officio member of the committee.
- A representative of the nursing staff (mostly senior nursing staff may include).
- A pharmacist. The pharmacist is designated as the “Secretary” of the PTC.
The members of PTC should meet regularly at least recommended six times a year and whenever is necessary. The committee can also call other persons which may be within or outside the hospital. The rationale for inviting such additional members is to the contribution of specialized or unique knowledge and skilled judgment in the meeting.
The agenda and the supplementary materials should be prepared by the secretary and should be furnished to the committee members sufficiently in time before the meeting. For example, the typical agenda for the meeting of PTC are as follows:
- Minutes of the last meeting.
- Review of specialized contents of hospital formulary for up-dating and deletion of some products. The purpose of this agenda is to bring the formulary up to date and delete the products which are no longer in use or consideration.
- Review of investigational drugs which are currently under processing in hospital.
- Review the side effects, toxic effects, drug interactions, adverse drug reactions reported by patients in units of hospital since the last meeting.
- Drug safety in the hospital.
The mission of PTC:
The mission of the Pharmacy and Therapeutic Committee is to encourage the health care professionals (physicians) for prescribing and recommend using high-quality and cost-effective medication for patient health care.
Goals and Objectives:
- To promote the appropriate use of high-quality and cost-effective medication.
- To ensure compliance with applicable standards and federal regulations.
Responsibility of Pharmacy and Therapeutic Committee:
- Preparation of hospital formulary.
- Ensure patient safety regarding proper medication.
- Broadcasting of bulletin.
- Formation of automatic stop orders for dangerous drugs.
- Regulation on the use of investigational drugs.
- Involved in the preparation of kits or carts for a medical emergency.
- Program development for the reporting and assessing of the adverse drug reactions.
Functions of The Pharmacy and Therapeutics Committee
The Pharmacy and Therapeutic Committee is responsible for the following major functions:
- To select and make availability of drugs by the concept of essential drugs.
- To recommend the procedures to procure the selected drugs and adopt the criteria and procedures for the procurement of drugs.
- To estimate hospital drug requirements and manage the hospital drug budget.
- To define standards for prescribing drugs and related practical requirements.
- To make availability of drugs to all prescribers and dispensers unbiased information on drugs.
- To organize information drives on drugs for the general public.
- To regulate the commercial promotion of drugs inside and outside the hospital.
- To inform all hospital staff members about all decisions taken by the pharmacy and therapeutic committee.
- To conduct scientific studies to update and ensure rational use of drugs.
- To investigate periodically the morbidity and mortality statistics to determine changes in their patterns, requiring changes in rational drug therapy.
- To monitor adverse drug reactions of drugs.
- To develop and implement dispensing procedures and practices.
- To design a strategy assuring the quality of the drugs and the pharmaceutical services in the hospital.
- To develop and undertake educational activities on the rational use of drugs.
- A committee has the power to make out and address conflict of interest issues that will obstruct the practitioner’s capability due to financial or some other reasons.
In general, the PTC should serve in an evaluative, educational, and advisory capacity to the medical staff and organizational administration in all matters about the use of medications.
Role of Pharmacy and Therapeutic Committee in Drug Formulary
The pharmacy and therapeutic committee is responsible for preparing and maintaining an updated formulary. The committee follows three main basic principles in the preparation of the formulary. These principles are based on the weightage of the proven effectiveness of the medication, the safety of the patients, and most important is the cost of medicine.
1. Proven effectiveness documented in the medical literature: The primary concern in the preparation of formulary is consideration of the degree to which a medication produces desirable clinical effects. Before inclusion of new medicine/drug in the list of formulary, PTC should assess and discuss the favorable effect of new medication based on its strength of scientific evidence in the reported literature, its pharmacoeconomic studies, case reports, randomized clinical trial, the outcome of the research and the medical opinion on the new drugs.
2. Maximizing safety and minimizing the potential for errors: During the preparation of the formulary, PTC should assess the risk and benefit ratio of the new product/medicine compared to the existing drug product in the treatments. During preparation, there is a need to minimize the errors that may cause by the appearance of the products in formulary such as; the name of drug product, dosage form, and packaging that carries fears to the safety of patient or potential for errors in the health care system.
3. Optimizing pharmacoeconomic: It is the responsibility of PTC to compare the overall cost of a new medicinal product with existing treatment costs as well as compare the risk and medical outcomes. Generally, PTC gives the preferences to those medicinal products whose safety and efficacy profile are much more compared to existing treatment at an affordable cost to the largest potential population.
4. Prominence of the medicinal products essential to health care.
5. Patient convenience, adherence, and satisfaction: PTC has prime responsibilities to review the favorable medicinal products to increase patient convenience, adherence, and satisfaction. Such as the inclusion of convenient dosing, various form of dosage forms, storage requirements, ability to divide the dosage by themselves.
6. To support the standard treatment algorithm while preparation of formulary.
7. Long-term stability of formulary decisions: PTC should be able to look towards the stability of formulary for a long time. For the stability of members of the committee, the agenda regarding changes in the formulary should be minimized.
8. The formulary will serve as a guideline for the vast majority of patients: To promote the utilization of formulary, the PTC should apply the management policy such as prior authorization, quality limits, step-wise edition in the formulary, and the age limit of the formulary. Considering the patient health care, the formulary should be readily available and easy to use promptly so such type of care should be taken by PTC.
Role of PTC in Dispensing of medication to IPD and OPD
PTC has the responsibility to establish the policy for drug distribution to inpatients and outpatients care services. In these services, before dispensing a drug, a pharmacist must make sure about the correct prescription of the drug and its validity with regards to diagnosis and treatment. Pharmacists should also check for any modification concerning the dose regimen.
PTC also supervises a steady supply of drugs as per the needs of the patients and health care partners. In this, IPD/OPD pharmacist works together with inventory, drug distribution in-charge, and physicians.
PTC has a contribution to managing and advisory role to a pharmacist who is working in the hospital. In this, PTC must guide pharmacists to supervise regarding proper distribution of drugs across inventory, pharmacy, floor pharmacy, ward pharmacy, IPD, OPD, etc. to avoid ambiguities or any other failures.
The PTC should also assist/guide the pharmacist regarding supervising the purchase orders, manage logs of material transfer across departments, and maintain smooth functioning of drug distribution across the hospital.
Role of PTC in Automatic Stop Orders for Dangerous Drugs
PTC has to set the policy for the automatic discontinuation of all medication prescriptions after 48 hours for sedative and hypnotics, narcotics, anticoagulants, and antibiotics-containing drugs. Another way is prescription strictly indicates the dispensing of an exact number of doses to be administered and if require more need to re-order the medications. There must be a policy of rewrite of prescription order every 24 hours for narcotics and CNS active drugs. While such type of practice is not routinely used in Indian hospitals except a few such as Juslok Hospital Mumbai, Mayo Hospital, and Christian Medical Hospital Vellore.
Role of PTC in Developing of Emergency Drug List
Considering the time factor in an emergency, the PTC of a respective hospital must get ready for the availability of emergency kits/drug boxes/medicine for patient use at the bed site. The list of such emergency drugs should be compiled by PTC and placed in an emergency kit. Following are listed some emergency drugs and supplies compiled by the PTC:
(A) Supplies to be maintained in Emergency Box:
Table.1: Emergency Supplies
| Sr. No. | Emergency Supplies |
| 1. | Syringes of the various range such as 1, 2, 5, 10, and 20 ml. |
| 2. | Needles of 16′, 18′, 20′, 21°, 23′, and 26′. |
| 3. | Airway Equipment. |
| 4. | Tourniquets. |
| 5. | Files. |
(B) Drugs for Emergency Box:
Following are the emergency drugs that are selected to keep only in consultation with a physician.
Table.2: Drugs for the Emergency Box
| Aminophylline | Diphenylhydantoin | Mannitol Injection |
| Atropine Sulphate | Epinephrine | Nalorphine |
| Caffeine | Heparin | Neostigmine |
| Calcium gluconate | Hydrocortisone | Saline Dextrose Injection |
| Digoxin | Isoproterenol |
(C) Supplies for Cabinet Utility Room:
Table.3: Supplies for Cabinet Utility Room
| Burn Sheet | Resuscitation | Tracheotomy set |
| Oxygen equipment | Suction catheter | Venous Catheter |
| Phlebotomy set | Sterile sponge and Gauge | Venous cannulation Set |
| Razor with blades | Tubing set |
Role of Pharmacy and Therapeutic Committee in Drug Product Defect
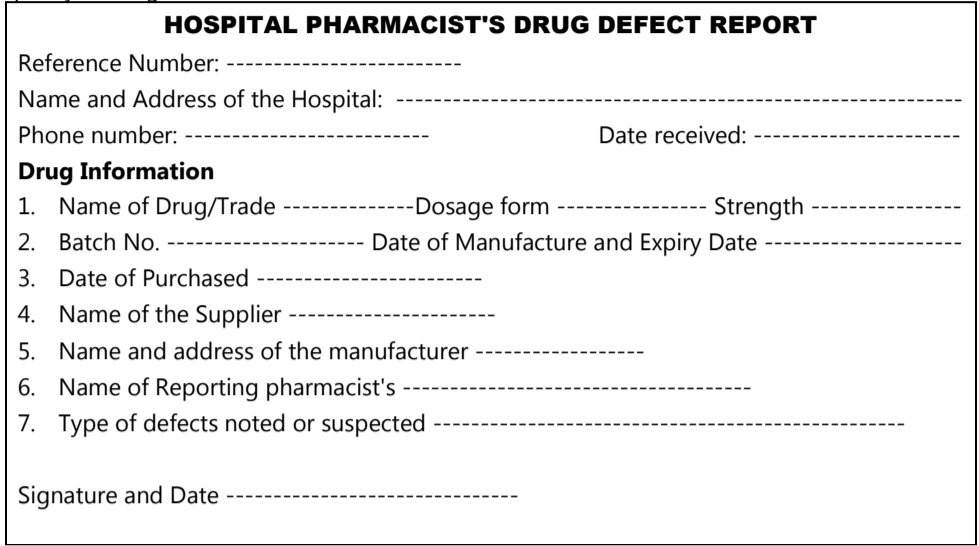
The drug purchase committee of the hospital may report to the PTC about the information about receiving the defective quality of the drug from the manufacturer. The PTC has a responsibility to inform first to manufacturer/supplier of necessary action. In case of unsatisfactory answers from the manufacturer or supplier, then report to the Food and Drug Control Administration. Following is the general format for reporting about the defective quality of drugs.
Make sure you also check our other amazing Article on : Therapeutic Drug Monitoring
